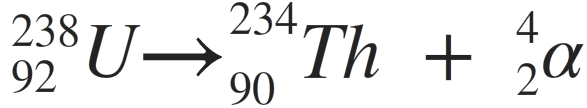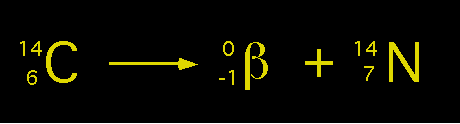Alpha Radiation
Alpha Radiation consists of helium nuclei that have been emitted from a radioactive source. Each of these emitted particles, called alpha particles, contain two protons and two neutrons and have a double positive charge.
An example of an equation: radioisotope uranium-238 releases alpha radiation and is transformed into another radioisotope, thorium-234
An example of an equation: radioisotope uranium-238 releases alpha radiation and is transformed into another radioisotope, thorium-234
The equation, shown above, is balanced. The sum of the mass numbers on the right equals the sum on the left. When an atom loses an alpha particle, the atomic number of the product is lower by two and its mass number is lower by four. Due to their large mass and charge, alpha particles do not usually travel very far and are not very penetrating; for example, a sheet of paper of the surface of your skin can easily stop them. Radioisotopes that emit these particles are dangerous when ingested.
Alpha radiation is composed of alpha particles with helium nuclei. its symbol is:
Alpha radiation is composed of alpha particles with helium nuclei. its symbol is:
it has a 2+ charge and a mass of 4amu. The common source of alpha radiation comes from Radium-226. Its penetrating power is low, only 0.05 mm of body tissue, and it can be shielded by paper and clothing.
Beta Radiation
A beta particle is formed when an electron breaks apart from a neutron in an atom. The neutron breaks apart into a proton, which remains in the nucleus. The symbol for the electron has a subscript of -1, representing the electron's negative charge, where the atomic number would be written. The superscript 0 where a mass number would be written represents the extremely small mass of the electron compared to that of a proton. A beta particle has less charge and much less mass than an alpha particle. Consequently, beta particles are more penetrating. Beta particles can pass through paper but are stopped by aluminum foil and thin pieces of wood.
Carbon-14 emits a beta particle as it undergoes radioactive decay to form nitrogen-14.
Carbon-14 emits a beta particle as it undergoes radioactive decay to form nitrogen-14.
The nitrogen-14 atom has the same atomic mass number as Carbon-14, but its atomic number has increased by 1. The equation is balanced because it contains an additional proton and one fewer neutron.
Beta Radiation is composed of Beta Particles, which are electrons. The symbol for this type of radiation is:
Beta particles have a 1- charge and a mass of 1/1837 amu. The common source of beta radiation comes from Carbon-14. Beta particles are able to penetrate 4mm of body tissue, which is moderate, but can be stopped by aluminum foil.
Gamma Radiation
A gamma ray is a high-energy photon, which is electromagnetic radiation, emitted by a radioisotope. Nuclei often emit gamma rays along with alpha or beta particles during radioactive decay.
Gamma rays have no mass and no electrical charge, therefore the emission of gamma radiation does not alter the atomic number or the atomic mass of an atom. Gamma rays are extremely penetrating and can be very dangerous. They pass easily through paper, wood, and the human body. They can be stopped, but not completely, by several meters of concrete or several centimeters of lead. The common source of gamma radiation is Cobalt-60.The symbol for gamma radiation is: